Get the case study as a PDF.
Support the Stratification of Antiviral Treatment After Allogeneic Hematopoietic Cell Transplantation
Background
Allogeneic stem cell transplant is a common treatment for many leukemias and other blood cancers. Transplant patients are particularly susceptible to high morbidity and mortality from cytomegalovirus (CMV) reactivation due to suppressing cell-mediated immunity following transplant.
Anti-CMV drugs can control CMV reactivation, but side effects are common, and at some point, the patients need to come off the drug. Long-term control of CMV reactivation is dependent on CMV-specific T cells. Determining a patient's CMV immunity can inform when the patient has immunity to fight the virus upon reactivation and consequently no need for antiviral treatment.
MHC Dextramer®
Study Description
A team at Roswell Park studied 315 patients who received hematopoietic stem cell transplants from an allogeneic donor between 2008 and 2015. The goal was to determine how many suffered from CMV reactivation and developed anti-CMV immune responses following transplantation.
The cellular immune responses to CMV were studied in the first 365 days after transplantation by detecting the presence and quantification of CMV antigen-specific CD8+ T cells (CAST) using MHC I Dextramer® reagents in peripheral blood samples by flow cytometry.
Of the predominantly European American patients seen at this transplant center, 87% had an HLA-A or B allele that matched the CMV-specific MHC I Dextramer® reagents used in this study.
Results
Early CMV reactivation might be protective since it causes the patient’s immune system to induce CASTs that may improve the ability to prevent subsequent CMV infection.
Specifically, the team’s results suggest that:
- A low-level of CMV infection before day +100 after allogeneic hematopoietic cell transplantation may be beneficial if CMV-specific immunity is reconstituted and may not require treatment until progression of CMV reactivation.
- Patients with high risk to reactivate CMV: recipient/donor (R/D) CMV serology of R+/D- and R+/D+ who reactivated CMV and developed ≥ 3 CAST/µl of blood were less likely to subsequently reactivate CMV (Fig. 1).
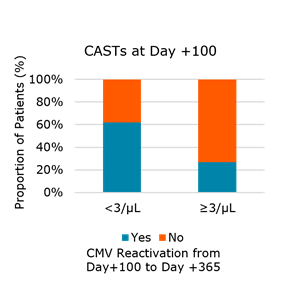
Fig. 1. Number of CMV antigen-specific CD8+ T cells (CAST) at day +100 correlates with subsequent CMV reactivation in R-D+ and R+D- transplant pairs that reactivated CMV before day +100 (P=0.4)
Conclusions
- It may be beneficial to allow patients, under careful supervision, to develop a low-level viral infection without immediately providing medication
- By testing patients for the presence of CAST, the medical teams can determine whether CMV could be controlled by the patient’s immune system alone without medication
- CMV Dextramer® reagents are sensitive tools to measure levels of CAST and access immune reconstitution

