CD22 CD7 GD2 EGFR Mesothelin
CD20 CD38 HER2 Claudin-18.2
CD30 Kappa light chain IL-12Rα2 PSMA
The market approval of Kymriah® in 2017 marks the beginning of a new era in biopharmaceutical development characterized by the transition of living cells from producing to being active therapeutic agents. In the last years, hundreds of patients have been treated with bioengineered T cells. Today, six commercial CAR-T cell therapies are available to treat seven hematological cancers1, while over 2000 clinical trials (200 in phase III) test investigative cell therapies2.
There is an important distinction between the handful of approved CAR-T cell therapies from the plethora making its way along the path from bench to bedside. The approved therapies are conspicuously homogenous: four target CD19 using the FMC63 scFv, and the other two target BCMA. All use CD3ζ as signal 1 and either 4-1BB or CD28 as the costimulatory signal 2. In contrast, the upcoming innovations in CAR-T therapies explore an extended range of targets and a wide variety of structures and signaling strategies.3 That distinction raises questions about the adequacy of current tools to characterize the onslaught of diverse therapy candidates, not the least of which are staining reagents.
CD22 CD7 GD2 EGFR Mesothelin
CD20 CD38 HER2 Claudin-18.2
CD30 Kappa light chain IL-12Rα2 PSMA
Say you’re developing a novel CAR-T cell therapy. How do you go about validating the functional expression of your CAR in target lymphocytes, characterizing the transduced cell population, or monitoring CAR-T cell populations in the blood over time? You could do it by measuring gene expression. However, to demonstrate the presence of the CAR on the cell surface, you will need protein staining reagents. Figure 1 summarizes the different types of protein staining reagents currently used to analyze CAR-T cells.
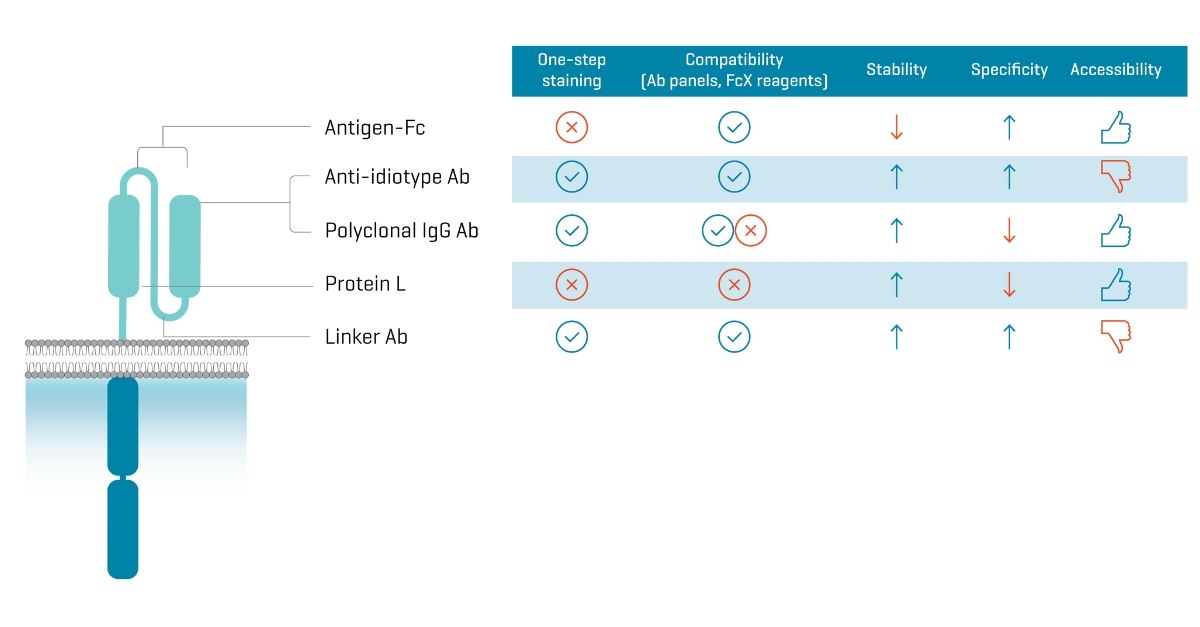
Figure 1. Features of protein staining agents currently used to characterize CAR-T cells. Binding locations on the extracellular domain of a CAR are labeled in the diagram on the left.
At first glance, options seem plentiful. However, availability and shortcomings narrow the selection. Two options are difficult to obtain. Linker antibodies are monoclonal antibodies developed by Kite Pharma for Yescarta® and Tecartus®, they are heavily engineered and specific for the CAR construct for which they were designed. Similarly, anti-idiotype antibodies are stable, specific, and versatile, however they are not commercially available and must be developed for each specific CAR construct of interest, at significant cost.4 Protein L is a bacterial protein that binds the light chain of scFv but can have a low staining index and very low power in discriminating transduced from untransduced cells depending on the CAR construct.5 Finally, polyclonal anti-IgG antibodies, though stable and easy to obtain, are not always compatible with blocking reagents and other antibodies used in phenotyping cells. Plus, like protein L, they show inconsistent performance in terms of specificity.5
That leaves you with antigen-Fc – chimeric proteins with an antigen (the target of your CAR) fused to an Fc fragment. The great advantage of these CAR staining reagents is that they are not only specific to the CAR construct itself but also to the antigen it targets. This is a decided advantage for next-generation bivalent and other gated CAR strategies where it is important to demonstrate the concurrent expression of two extracellular domains. Antigen-Fc reagents are accessible, compatible with additional antibody staining, and newer reagents are conjugated with fluorophores to perform one-step staining.
FDA Guidance for Industry recommends the direct detection of the CAR to determine the percentage of CAR-positive cells. Dextramer® reagents may provide a sensitive solution for direct detection and quantification of CAR-positive cells by flow cytometry.
Where antigen-Fc reagents can fall short is in sensitivity. With a single valency for binding, these reagents suffer from the same limitations as MHC monomers. Multivalent interactions with CAR constructs on a transduced T cell decrease their dissociation rate for a longer residence time on the cell. Hu et al. examined the sensitivity of monomer antigen-Fc in detecting 3 different CAR constructs compared to multimer reagents. Titrations showed that the multimer reagents had significantly higher sensitivity, most dramatically for CAR constructs with high dissociation rates for their antigen ligand.5
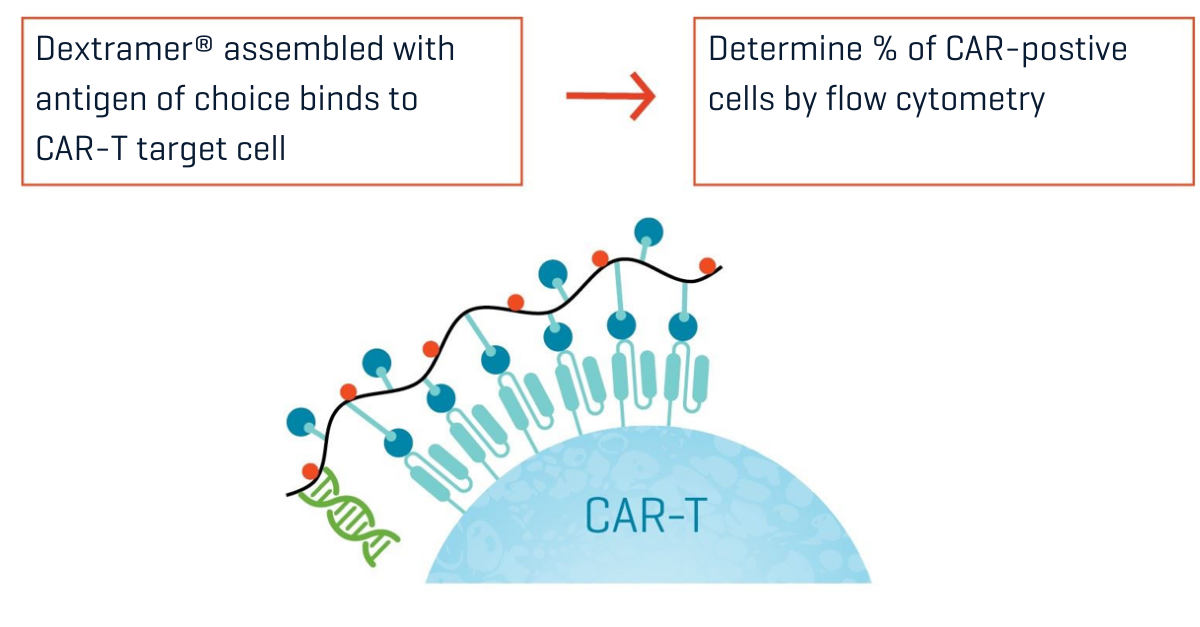
Figure 2. Creation of Antigen Multimers using Dextramer® technology. Dextramer® reagents may provide a sensitive solution for direct detection and quantification of CAR-positive cells by flow cytometry.
With their dextran backbone displaying multiple binding sites, Immudex reagents are a clear example of the advantage that comes with the high avidity of multivalent binding. MHC I Dextramer® and MHC II Dextramer® reagents are used by researchers around the world because they detect low-affinity antigen-specific T cells that are invisible to corresponding tetramers. The same Dextramer® technology can be loaded with biotinylated antigens to create a powerful antigen-specific CAR staining reagent (see our poster “Antigen-Specific Solutions for T-Cell Therapy Development and Manufacturing”). The dextran backbone also accommodates multiple fluorophores to generate a high signal intensity, and reagents can be designed with different fluorochromes. This opens the option of multiplexed staining to detect the presence of complex constructs, like bivalent or gated CARs.
Two other factors heighten the importance of using high-avidity reagents beyond the need to detect low-affinity CARs. First, transduction efficiency varies widely depending on CAR construct. With low transduction rates, it is imperative to boost sensitivity so low-abundance CAR-T cells don’t go undetected. Second, expression rates can vary as well. When cells display fewer CARs, multivalent staining reagents, like those built on a dextran backbone, are more likely to bind them.
Dextramer® reagents have one additional feature that makes them a superior choice for CAR-T cell detection and analysis: They can be combined with DNA barcodes to enable NGS-based discrimination of different population subsets or single-cell multi-omics, all from a single sample. You leave no cell undetected, and no cell unidentified.
So. What’s in your cytometer?
Explore how Dextramer® reagents support the development of effective cell therapies.
Learn how Dextramer® technology may enable detection and characterization of CAR-T cells.
Our team can help you develop custom Dextramer®-based solutions for CAR-T detection.
Fill in the form and one of our dedicated specialists will get in touch with you shortly.