Get the case study as a PDF.
Assessing HLA-Specificity of CAR-T Cells with MHC I Dextramer®
Background
Human regulatory T cells (Tregs) can be efficiently engineered using chimeric antigen receptors (CARs) targeting donor-specific antigens, thus enable more potent immunosuppression compared to polyclonal Tregs in preclinical models of transplantation. CAR engineering also allows generating cells that express other molecules for the optimized production of CAR-Tregs.
This study provides a proof-of-principle for engineering human CAR-Tregs with certain HLA-specificity co-expressing IL-10 and the application of Dextramer® technology in the next-generation CAR-Treg therapy.
Study Description
Goal: to examine the impact of IL-10 co-expression in engineered HLA-A2-specific CAR-Tregs.
Different CAR-Tregs were generated by lentiviral transduction of 4 different constructs: CAR specific to HLA-A2 alone (A2-CAR), IL-10 alone (IL10-poly), both constructs (IL10-A2-CAR), and neither construct (Poly). The transduction efficiencies were assessed by NIS-TRFP expression by FACS. Transduced cells were further expanded and assessed for phenotype and suppressive function.
To quantify the CAR expression, Tregs were stained with MHC I Dextramer® specific for HLA-A2 /CINGVCWTV using flow cytometry, whereas IL-10 expression level was assessed by ELISA.
Results
- Both IL10-A2-CAR-Tregs and A2-CAR-Tregs were confirmed to express HLA-A2-specific CAR by positive staining with MHC I Dextramer® (Fig. 1A).
- Both IL10-A2-CAR-Tregs and A2-CAR-Tregs upregulated CD69 after co-culture with the A2+ B-LCLs (49.9 ± 4.6% and 54.9 ± 5.9%, respectively) but not after co-culture with the A2− B-LCLs (Fig. 1B).
- Human Tregs lacking the A2-CAR had no significant CD69 upregulation after co-culture with A2+ B-LCLs.
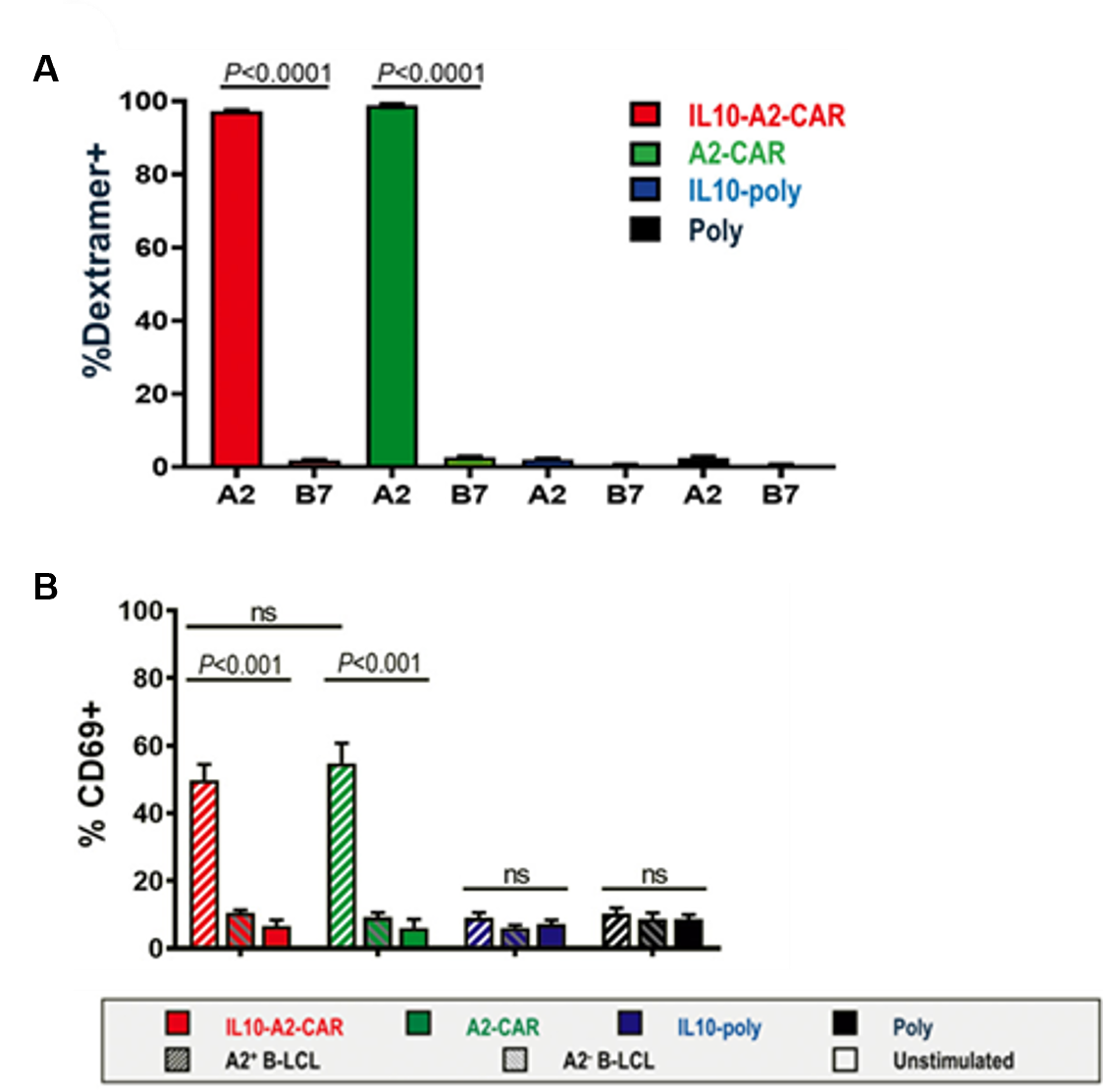
Fig.1. A. HLA-A2/CINGVCWTV and HLA-B7/APRGVRMAV (control) MHC I Dextramer® were used to quantify CAR surface expression by flow cytometry. B. Antigen-specific Treg activation was assessed by CD69 upregulation. Transduced Tregs were cultured with indicated B-LCLs for 18h followed by staining with anti-CD69 antibody and analysis by flow cytometry.
Conclusions
- Human A2-CAR-Tregs co-expressing IL-10 maintained a stable phenotype and were specifically activated by HLA-A2.
- CAR technology can be efficiently applied for generating Tregs that are HLA-specific.
- Dextramer® technology is a valuable tool for monitoring the presence of antigen-specific CARs in engineered CAR-T cells.

