Get the case study as a PDF.
In-Situ Immune Cell Infiltrate and Immunoscore are Useful Indicators of Tumor Recurrence and Survival
Background
Tumors can be classified based on multiple morphologic, phenotypic, and functional factors, including microsatellite instability (MSI). MSI in colorectal cancer (CRC) predicts favorable outcomes, which has been related to its effect on the antitumor immune response.
This study assesses the immune status via Immunoscore as a potent indicator of tumor recurrence beyond MSI staging that could be an important guide for immunotherapy strategies.

Study Description
Goal: to characterize the existence of antitumor T cells within CRC tumors, in relation to tumor genotype, instability status, and immune response.
A comprehensive analysis of mutation patterns, chromosomal instability status, RNA sequencing data, tissue immune responses, Immunoscore, and their effect on overall survival (OS) was done in two cohorts of CRC patients in relation to MSI or microsatellite-stable (MSS) tumors:
- cohort 1: 270 CRC patients from the TCGA dataset (Cancer Genome Atlas Network, 2012)
- cohort 2: 694 tissue samples from CRC patients with primary resection of their tumor.
Tumor-infiltrating CD8+ T cells were detected by in-situ staining with HLA-A*0201 MHC I Dextramer® reagents. The correlation between MSI, Immunoscore, and patient survival was further investigated.
Results
- MSI tumors had increased frameshift mutations, with frequent TGFBR2 frameshift mutations in MSI, but not MSS, tumors (data not shown)
- MSI tumors had higher densities of in-situ proliferating T cells compared to MSS tumors and were infiltrated with TGFβR2-specific cytotoxic T cells (Fig.1)
- MSI patients had high infiltration of effector memory cells and high Immunoscore (data not shown)
- Patients with low Immunoscore had shorter OS than patients with a high Immunoscore, regardless of MSI staging (data not shown).
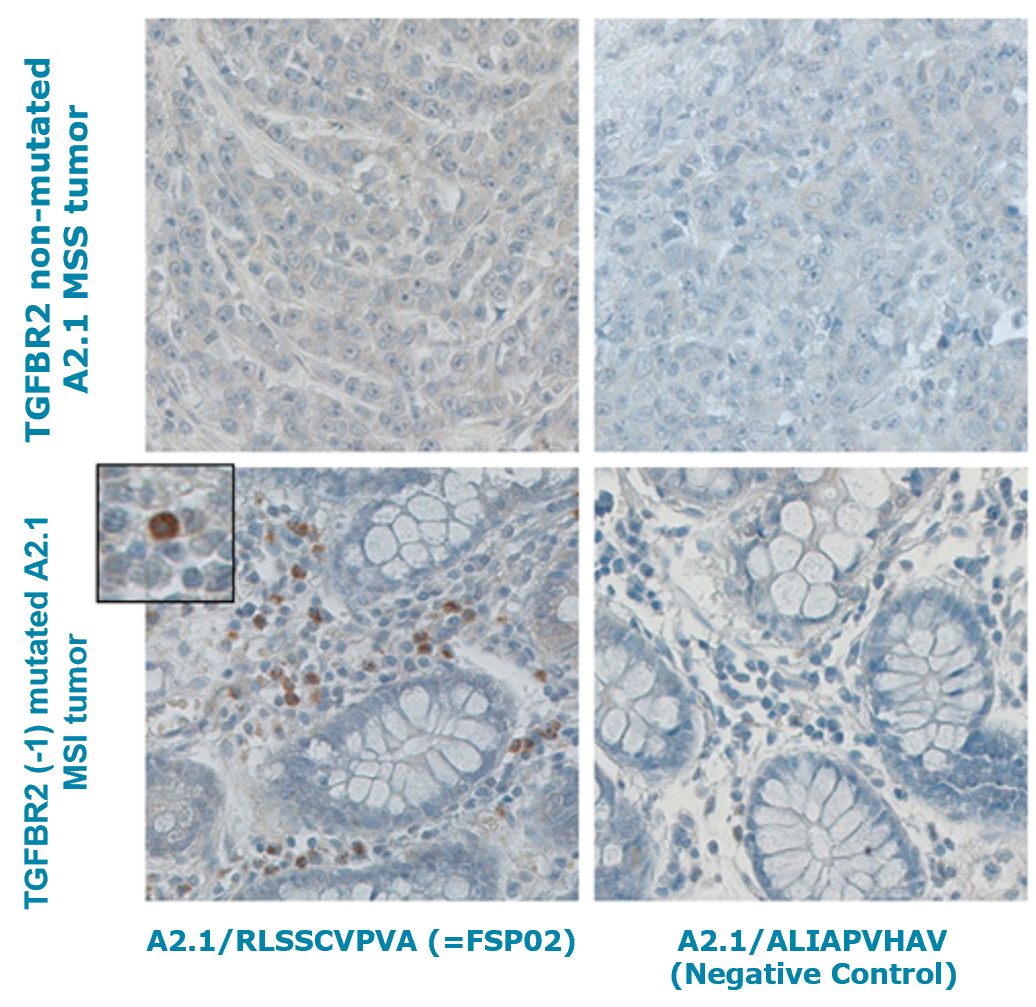
Fig.1. HLA-A2/peptide immunohistochemistry analysis in FFPE sections of colorectal cancer patients using HRP/peroxidase enzyme for detection. In-situ staining toward TGFBR2 (-1) mutation-derived neo-antigen FSP02 with MHC I Dextramer® FITC HLA-A*02:01/RLSSCVPVA (FSP02). MHC I Dextramer® FITC HLA-A*02:01/ALIAPVHAV served as a negative control.
Conclusions
- High-frequency frameshift mutations in MSI might affect the clinical outcome by boosting the presence and functionality of tumor-infiltrating CD8+ T cells in CRC patients
- A tumor-specific T-cell response toward a mutated TGFBR2 class I neo-epitope could be detected in the primary tumor with MHC I Dextramer® for In-Situ Staining reagents
- Immune infiltration and Immunoscore are an indicator of patient survival, regardless of MSI.

