Get the case study as a PDF.
Plasmacytoid Dendritic Cell Line-Based Vaccine Primes and Expands Antitumor T Cells in Melanoma Patients
Background
The immune checkpoint inhibitors (ICI) are the golden standard treatment for metastatic melanoma, the efficacy of which depends on preexisting antitumor immunity. Plasmacytoid dendritic cells (PDC) are potent inducers of antitumor CD8+ T-cell responses and represent promising vaccine candidates that can be used in combination with ICI.
In metastatic melanoma patients, this first-in-human trial investigates antitumor immunity induced by the novel allogeneic PDC line based-vaccine (GeniusVac-Mel4).
Study Description
Goal: to assess the safety of the GeniusVac-Mel4 vaccine and its ability to induce antitumor immune responses in metastatic melanoma patients.
9 HLA-A*0201 patients with metastatic stage IV melanoma were split into 3 groups according to the vaccine dose received of irradiated PDC loaded with 4 melanoma antigens: MLANA (ELAGIGILTV), MAGEA3 (YMDGTMSQV), PMEL (FLWGPRALV), and TYR (IMDQVPFSV). Anti-vaccine immune responses were assessed in blood and metastasis by MHC I Dextramer® staining against melanoma antigens and further characterized in terms of naïve/memory profile by flow cytometry.
Results
- A significant increase in the frequency of circulating MLANA-specific CD8+ T cells was observed in 2 patients, accompanied by a switch from naïve to memory phenotype (Fig. 1).
- The analysis of metastasis by flow cytometry showed that MLANA-, PMEL-, and BMLF1-specific CD8+ T cells were highly enriched compared to the blood (data not shown).
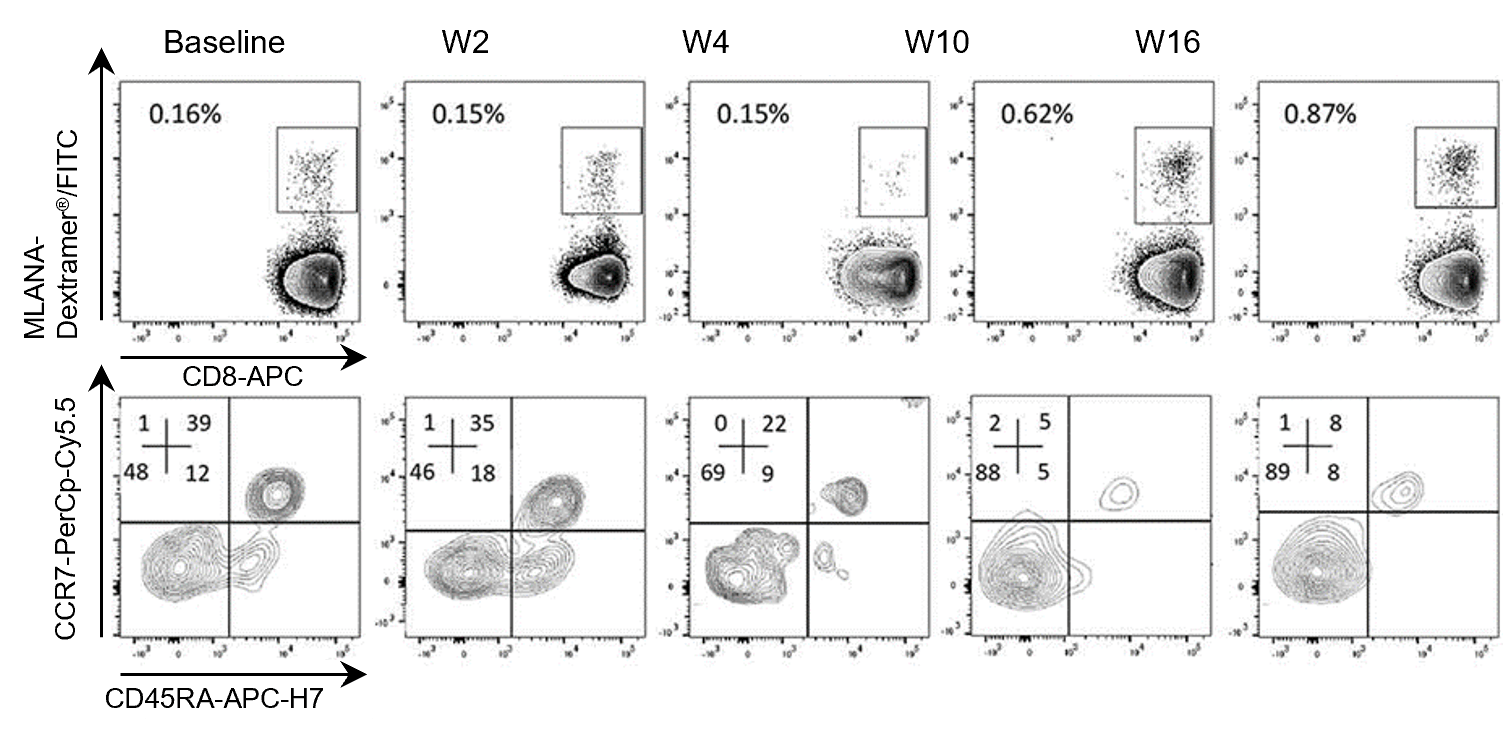
Fig.1. Immunological responses in a melanoma patient treated with GeniusVac-Mel4. Immune monitoring on fresh PBMC showing anti-MLANA T-cell percentages among CD45+CD3+CD8+ T cells in patient 0401 (group 2: 20×106 cells/injection). Specific T cells were analyzed by flow cytometry, with MHC I Dextramer® labeling and characterized in terms of naïve/memory profile, according to their differential expression of CCR7 and CD45RA.
Conclusions
- A novel PDC line based-vaccine is capable of priming and expanding anti-tumor CD8+ responses in cancer patients.
- Melanoma-specific MHC I Dextramer® reagents can be used to monitor vaccine-induced T-cell populations in patients’ blood and metastasis.
- Future studies require testing the combination of the PDC line vaccine with immune checkpoint inhibitors.

